Featured Article

One goal of mammalian upstream bioprocess development is to deliver a process that is safe and robust and that produces enough biopharmaceutical material to meet demands. Process development teams are increasingly asked to do more and in less time; automated feeding strategies are thus becoming more widely used to streamline work flow. They have also been shown to improve process performance and product consistency.
The primary feed component of cell cultivation is glucose, which is used as a starting point for all growth and energy pathways within cells. Ensuring that the cells have the right amount enables them to grow rapidly and maximize product secretion. Excess glucose concentration has an effect on the glycosylation rate of secreted soluble proteins. Thus, controlling the available glucose improves the consistency of glycosylation patterns on final products and may improve quality.
Material and methods
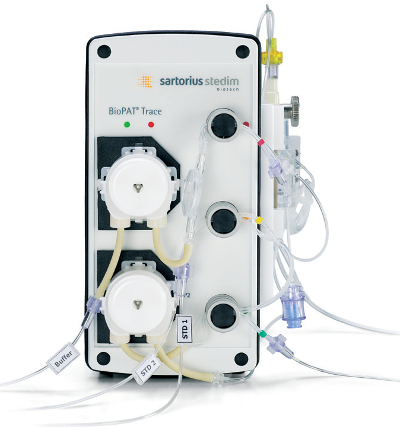 Figure 1 – BioPAT Trace.
Figure 1 – BioPAT Trace.This article describes a method used to establish glucose feed control using Sartorius Stedim Biotech (Goettingen, Germany) hardware and equipment. The BIOSTAT B-DCU bioreactor for advanced process optimization and characterization offers the option of a glucose concentration controller. Integration of the BioPAT Trace (Figure 1) allows real-time monitoring of the glucose level, enabling a user-defined glucose setpoint within the bioreactor’s control software. In the first two runs, in situ glucose concentration was maintained by discontinuous bolus feeds. The third run utilized a glucose setpoint controller using defined PID settings and an internal speed-controlled peristaltic pump.
The BioPAT Trace was configured for dialysis mode glucose and lactate measurement at a 20-min sampling rate. It was set to a fully automated and self-calibrating protocol as part of the schedule tab function. Each 17-day run used 12 L of transport buffer, two calibration solutions (high: 10 g/L glucose; low: 1 g/L glucose), and a 10-L waste container attached to the tube set. The dialysis probe was prepared, filled, and installed in the UniVessel Glass 5 L prior to sterilization and was connected to the BioPAT Trace tube set and primed for analysis before inoculation (Figure 2).
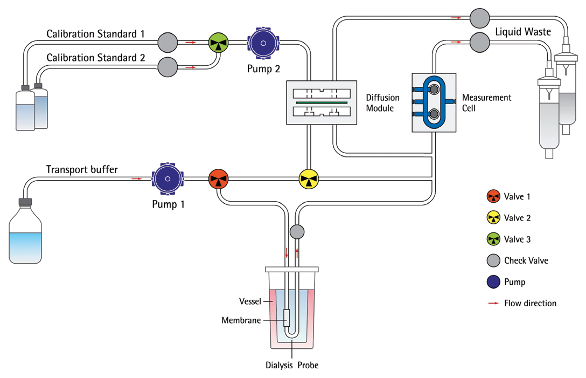 Figure 2 – Working principle of BioPAT Trace.
Figure 2 – Working principle of BioPAT Trace.The UniVessel Glass 5 L was equipped with two 3-blade segment impellers for low shear stress and homogenization of the cell broth. Blade angle was 30° and set to down-pumping. A ring sparger with holes facing up was used for all trials. The vessel was equipped with several ports for feeding, a classical pH sensor, pO2 sensor, dialysis probe, exhaust cooler, and gas filters.
A Chinese hamster ovary (CHO) fed-batch process was used to evaluate BioPAT Trace integration in the BIOSTAT B-DCU. The 17-day cultivation comprised a three-day batch phase followed by a 14-day fed-batch phase. After inoculation with 0.3 × 106 cells/mL, the peak viable cell density (VCD) was typically reached at day 8 with 25–30 × 106 cells/mL and a viability of 99%. After the following nine-day death phase, the VCD was above 10 × 106 cells/ mL, with a viability of more than 50% at the point of harvest.
Bolus feeding from day 3 was feed medium A (FMA), feed medium B (FMB), and a highly concentrated glucose solution (400 g/L). The amount of FMA and FMB was kept constant throughout the complete fed-batch phase. On day 7, additional glucose was needed to maintain a glucose concentration of at least 3 g/L in the cell broth. The feeding process was automated using balances and pumps connected to the digital control unit (DCU) and S88 set in BioPAT MFCS SCADA software. Offline glucose and lactate measurements were performed with an ABL800 basic (Radiometer America, Brea, CA). VCD and viability were analyzed with a Cedex HiRes analyzer (Roche, Indianapolis, IN).
Three trials were performed:
1) Monitoring—Trace technology was used to monitor the online glucose trend throughout the cultivation.
2) An online value replaced offline measurement. During the CHO culture, a sample was taken daily. The offline glucose value was used to fill to a certain glucose concentration in the bioreactor. In trial 2, the glucose online value was used instead of the offline value for this feeding procedure.
3) Glucose control—during the third trial, glucose control at 6 g/L was activated after six days. The FMA bolus feed was modified to continuous feeding to reduce glucose concentration peaks.
The feeding scheme for trials 1 and 2 is shown in Figure 3 and was identical. During the batch phase, no medium was fed to the culture. On day 3, a daily bolus feed of FMA and FMB was started. After glucose fell below 6 g/L, an additional feed of glucose (400 g/L) was initiated. In trial 3, FMA bolus feeding was modified to continuous feeding to remove daily peaks in the glucose concentration (Figure 4). After glucose reached a concentration below 6 g/L, a controlled feed of glucose solution (400 g/L) was started to maintain the glucose concentration at 6 g/L.
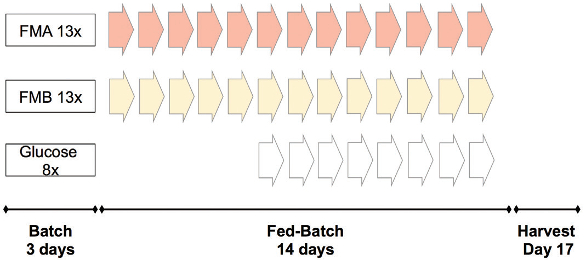 Figure 3 – Feeding schemes 1 and 2.
Figure 3 – Feeding schemes 1 and 2.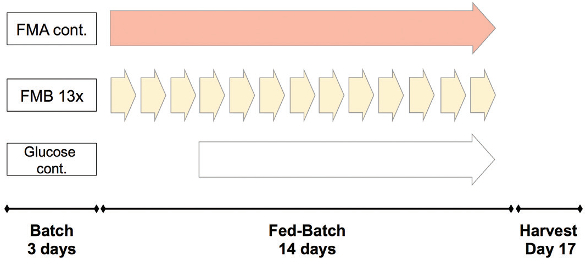 Figure 4 – Feeding scheme 3.
Figure 4 – Feeding scheme 3.Results and discussion
The batches ran sequentially on the same BIOSTAT B-DCU and BioPAT Trace systems. Results, shown in Figures 5 and 6, demonstrate the reproducibility of the process batches (compared to the historical “golden batch”). Glucose monitoring of the BioPAT Trace and subsequent glucose controller is shown in Figures 7–9.
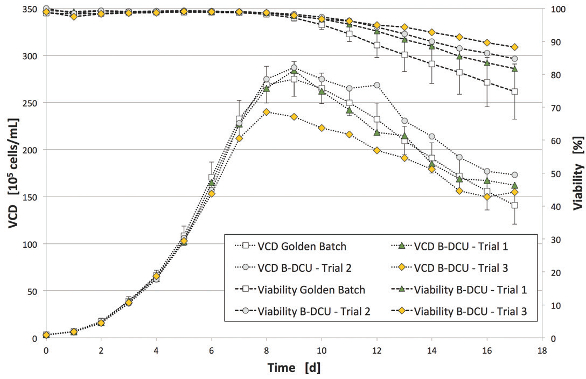 Figure 5 – VCD trend trial 1–3.
Figure 5 – VCD trend trial 1–3. Figure 6 – IgG trend.
Figure 6 – IgG trend.
Figure 7 – Glucose and lactate trend trial 1.
 Figure 8 – Glucose and lactate trend trial 2.
Figure 8 – Glucose and lactate trend trial 2.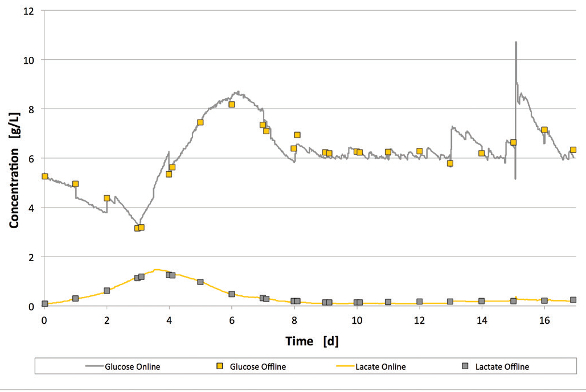 Figure 9 – Glucose and lactate trend trial 3.
Figure 9 – Glucose and lactate trend trial 3.Trials 1 and 2 fit the golden batch trend well. Due to a general process modification (continuous FMA feed), the VCD trend of trial 3 has a slightly reduced peak. Final VCD and viability exceeded golden batch values for all trials. Comparable productivity in all three trials was demonstrated by daily measurements of the product concentration (IgG concentration, Figure 6). Due to reduced cell growth in trial 3, a slightly lower product concentration was achieved in this run. Despite a small offset in the first 24 hours, the online and offline measurements of glucose and lactate fit well during the cultivation.
In trial 2, the online and offline glucose and lactate measurements were comparable; in trial 3, the online and offline trends fit well for glucose and lactate. At days 13 and 15, an unnecessary recalibration was performed (operational mistake).
Transition from glucose monitoring to glucose control requires alteration of the process. The effects of tighter glucose control are known to be beneficial to process performance and product quality.1 Considering this, it is not pragmatic to change a process using an untried, untested technology. Learning the system and building confidence around the measurement output (comparing to offline sampling) and ensuring the device’s robustness is generally needed before making a change. The first two process runs show the high-resolution monitoring capabilities of the integrated BIOSTAT B-DCU and BioPAT Trace, while the third run used the glucose controller. User interaction with the bioreactor as related to sampling glucose is not required, the time needed to maintain the BIOSTAT B-DCU system in a glucose-controlled state is reduced, and the process is more manageable (see Table 1).
Table 1 – FTE (full-time employee) resource balance per batch when using the BioPAT Trace for glucose

Conclusion
Stepwise integration of glucose control technology to a CHO fed-batch process was successful and maintained an in-process steady-state glucose concentration of 6 (±0.25) g/L. Integration of BioPAT Trace technology into the BIOSTAT B-DCU saves operator time and is an effective tool for improving product quality.
Reference
- Zhang, A.; Tsang, T. et al. Advanced process monitoring and feedback control to enhance cell culture process production and robustness. Biotech & Bioeng. 2015, 112(12), 2495–2504.
The authors are with Sartorius Stedim Biotech GmbH, August-Spindler-Strasse 11, 37079 Goettingen, Germany; tel.: +49 551 308 2487; e-mail: [email protected]; www.sartorius.com