Featured Article

The biopharmaceutical industry exercises great care to ensure the long-term stability, efficacy, and safety of therapies, from formulation development to patient administration. This includes selecting appropriate packaging materials during the product development and manufacturing process. A variety of container materials—from several types of glass and plastics to fusion materials—is available for parenteral drugs.
While manufacturers carefully consider such factors as the intended application of the therapy, possible interactions with the container, and drug properties when making a final material choice, they frequently overlook a crucial aspect of using containers made of new materials: micro- and nanoparticles. Yet, ignoring the subvisible particulates that can make their way into the formulation may jeopardize the success of an otherwise impeccably produced therapy. More education and discussion of the subject is needed to put nanoparticles in the limelight.
Invisible to the eye, dangerous to the body
The number of parenteral drugs has been steadily increasing over the past three years. For example, of the 35 new molecular entities approved by the FDA as of October 31, 2017, 14, or 40%, were parenteral drugs. The percentages of parenterals were, respectively, 50% in 2016 and 44% in 2015.1 Biologics in particular hold much potential for the biopharmaceutical industry, especially in light of the downward market pressures such as restricted access to high-cost branded therapies and generic competition. All of this prompts interest in advanced drug-delivery systems made of new materials offering convenience and ease of administration.
Subvisible particle detection and identification are particularly important for biologics, which are injected intravenously, subcutaneously, and intravitreally. These particles may include foreign contaminants (such as fibers, hair, and plastic), intrinsic particles from within the formulation that are capable of change over time (silicone oil), or inherent particles derived from the formulation itself (protein aggregation. (Figure 1). They can cause immunogenetic responses in patients due to unexpected interactions.2
 Figure 1 – Various protein agglomerates formed from individual, proteinaceous particles. Agglomerates can pose a threat to the safety, efficacy, stability, and longevity of parenteral bioformulations.
Figure 1 – Various protein agglomerates formed from individual, proteinaceous particles. Agglomerates can pose a threat to the safety, efficacy, stability, and longevity of parenteral bioformulations.Submicron-sized particles can agglomerate during various stages of the post-production process. One study showed that nano- and microparticles were formed during the peristaltic pumping process. During post-pumping agitation, there was a substantial increase in microparticle concentration coinciding with a significant depletion of the nanoparticle concentration, suggesting that agglomeration is the likely mechanism for larger microparticulate formation.3
Identifying subvisible particles is an important step in preventing recalls of parenterals, which, regrettably, happen frequently despite the rigorous process control typical of the industry. A notable case in point is the recall of Omontys, an injectable anemia drug recalled after seven people died in 2013 from serious allergic reactions. The FDA subsequently discovered that the formulation released to the market had a high concentration of subvisible particles. While the FDA did not establish a causal relationship between the particulates and the rate of anaphylaxis, the case highlights the importance of characterizing a drug product’s composition on the subvisible level.4
Reasons for the nano-disconnect
Most manufacturers follow the United States Pharmacopeia (USP) 788 guidelines for particle bin sizing in microns. USP 788 was designed to control for particles capable of causing capillary occlusion and focuses on those larger than 10 and 25 microns. It does not require any characterization of particles below 10 µm.5 Therefore, the current protocol literally overlooks subvisible particles and leaves a wide gap in assessing drug substance and drug product stability of parenteral therapies, to the peril of both biopharmaceutical drug makers and their patients.
According to USP 788, manufacturers must test parenteral formulations for subvisible particles using light obscuration and manual microscopy methods. The standard light obscuration method for particle analysis converts particle size and shape to that of an equivalent sphere by measuring the diameter of the shadow cast by the light source. This is not the most accurate way to measure size because the measurement is based on an inferred size, versus the actual size derived from the particle image. Standard nonimaging techniques, such as light obscuration, do not capture translucent particles as effectively as flow imaging technologies because the background of the particle is generally the same opacity as that of the translucent particle. For this reason, light obscuration is generally less accurate in counting and measuring protein particles that tend to be highly translucent.
While valuable to determine the count and concentration of particles greater than 10 µm, nonimaging techniques do not describe the particle’s morphology and its chemical characteristics. However, it is the morphology of the particle that allows scientists to determine its nature—inherent, intrinsic, or extrinsic. Morphology characteristics can include length, width, area, diameter, circularity (Hu), elongation, convexity, and others (Figure 2).
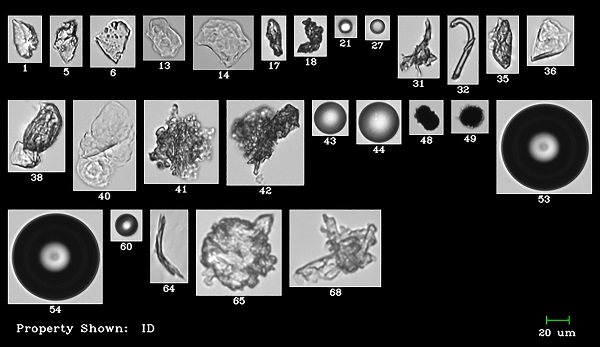 Figure 2 – The morphology of various types of particle—extrinsic, intrinsic, or inherent—allows biopharmaceutical scientists to identify specific contaminants and where they may originate during formulation development and process manufacturing.
Figure 2 – The morphology of various types of particle—extrinsic, intrinsic, or inherent—allows biopharmaceutical scientists to identify specific contaminants and where they may originate during formulation development and process manufacturing.Identifying contaminants is key to locating the problem in the process, as they can be introduced anywhere in production—from formulation development, quality assurance, fill finish, or storage, through transportation—and it is critical to remediate the issue in a timely manner. Therefore, the USP 788 protocols are no longer sufficient, particularly as manufacturers can now easily detect particles under 10 µm. Technology for the detection and imaging of nanoparticles has evolved significantly in recent years. One method is flow imaging, which allows for a more complete assessment of the nature and particulate load of protein therapeutics.
The regulatory and commercial winds of change
Industry experts have taken note of the need for comprehensive subvisible particle characterization. The FDA now recommends that flow imaging data and images be submitted with any new drug approval application. Moreover, a USP Expert Committee is currently at work to update USP 788 to include flow imaging as an orthogonal approach to complement light obscuration and manual microscopy.
While the FDA and USP are working on updating the standards, the industry needs to self-regulate and adopt innovative methods of flow imaging microscopy to detect particles down to about 300 nm. With it, scientists can detect protein agglomerates, silicon oil droplets, and other opaque, transparent, and translucent subvisible particles. Importantly, these high-resolution images provide particle morphology details in real time, allowing scientists to identify them.
Conclusion
Biopharmaceutical manufacturers strive to ensure patient safety, avoid recalls, and protect company reputations. Adequate particle analysis methods of packaging and drug-delivery materials should be part of their quality assurance and control effort. While new materials, such as plastic, solve certain problems of the past, more diligence is needed to identify inherent particulate matter in the new materials during the drug formulation and production process. Compliance with only the minimum requirements means that nanoparticles can be overlooked. Missed particulates can impact the therapeutic potency or alter the formulation with potentially disastrous consequences for the patient.
References
- https://dcatvci.org/4793-parenteral-drugs-tracking-new-drug-approvals
- https://onlinelibrary.wiley.com/doi/full/10.1002/jps.23973
- https://www.aiche.org/conferences/world-congress-on-particle-technology/2018/proceeding/paper/54bm-flowcam-nanor-provides-counts-sizes-and-images-nano-and-microparticles-application-therapeutic
- http://jpharmsci.org/article/S0022-3549(15)00180-X/abstract
- USP <787> Subvisible Particulate Matter in Therapeutic Protein Injections. Pharmacopeial Forum 39(2).
Kent Peterson is president and CEO, Fluid Imaging Technologies, Inc., 200 Enterprise Dr, Scarborough, ME 04074, U.S.A.; tel.: 207-289-3200; e-mail: [email protected]; www.fluidimaging.com