Featured Article
The evolution of the use of cannabis products has led to a growing number of specialized cannabis testing laboratories to provide the quality control needed to meet legislative requirements and protect consumer health and safety. Accurate measurement and reporting of the active ingredients in a product are essential for users to make their own evaluation and to ensure they have a reliable and predictable dose.
With high sample throughput and rigorous legislative requirements, laboratory information management systems (LIMS) are an extremely powerful tool for the testing laboratory. They allow all test data to be kept within a single, controlled database, providing a defensible QA framework for local legislative monitoring needs and many other benefits for the laboratory. In 2016, the use of a LIMS helped TEQ Analytical Laboratories (Aurora, CO) become the first ISO 17025-accredited recreational cannabis testing laboratory in North America.
Analyzing cannabis-based products
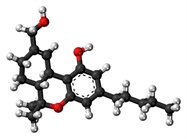
Cannabis contains multiple cannabinoids and other compounds. Although the use of the cannabis plant is not approved by the FDA, there are two FDA-approved drugs in pill form that contain a synthetic version of delta-9-tetrahydrocannabinol (THC) that occurs naturally in cannabis. These are used, for example, to treat nausea caused by chemotherapy and to increase appetite in patients with extreme weight loss caused by AIDS. Recreational marijuana-infused products (MIPs) include consumables such as cookies, candies, and beverages and topical applications such as lotions and bath soaks. It is the THC (Figure 1) in these products that is responsible for the high generally associated with marijuana.
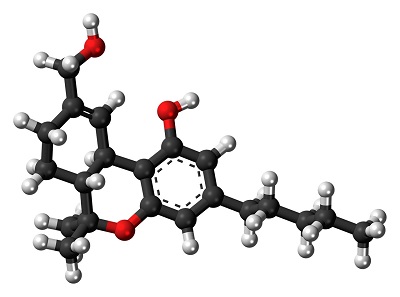 Figure 1 – Tetrahydrocannabinol.
Figure 1 – Tetrahydrocannabinol.Typical laboratory testing of raw materials and finished products includes analysis of:
- Cannabinoid potency
- Residual solvents
- Microbiological contaminants
- Yeast and mold
- Pesticides and herbicides
- Heavy metals
- Shelf life.
Testing is carried out in accordance with the requirements of the particular regulatory authority. For example, the Marijuana Enforcement Division (MED) is responsible for regulating the commercial marijuana industry in the State of Colorado.1 Licensed growers, manufacturers, and producers of edibles in Colorado are required to test their products for potency of five different cannabinoids—THC, THCA (tetrahydrocannabinolic acid), CBD (cannabidiol), CBDA (cannabidiolic acid), and CBN (cannabinol). Similarly, it is critical to ensure that residual solvents such as butane, heptane, benzene, toluene, hexane, total xylenes, as well as other solvents used during the extraction of cannabinoids from plant materials do not contain any hazardous or volatile compounds that could harm the consumer. Colorado also requires testing for three microbiological contaminants—Shiga toxin-producing Escherichia coli (STEC), Salmonella (Sal), and yeast and mold. Yeast and mold are normally used as an indicator for other contaminants and as a gauge for the general overall cleanliness of a product. Heavy metals testing is required in Colorado for lead, cadmium, arsenic, and mercury. While testing of the final product itself is essential, testing of the raw materials, plant materials, and other ingredients arriving at a manufacturing facility can prevent contamination in the final product.
Shelf-life studies measure the performance of a product over time. Information generated, such as the long-term stability of cannabinoids and terpenes in a product, can help determine an expiration or “best by” date. These studies can be tailored and independently derived based on the product’s ingredients, cooking processes (if appropriate), preservatives, and other factors.
The use of LIMS and ISO/IEC 17025
Accreditation to the ISO/IEC 17025 standard certifies that laboratories have the management, quality, and technical systems in place to ensure accurate and reliable analyses and proper administrative processes to confirm that all aspects related to the sample, analysis, and reporting are standardized, measured, and monitored. LIMS is a well-established tool used to control, manage, organize, document, analyze, and report information in QC laboratories and is therefore a natural adjunct for ISO 17025 compliance. At the testing level, a LIMS can be used to:
- Log incoming samples and print and scan barcodes to avoid errors
- Maintain complete chain-of-custody; track samples from initial sampling location through the lab to disposal; record location, custodians, and other key data
- Facilitate and organize laboratory data
- Account for the entire quantity of sample received, used, and disposed of
- Create certificates of analysis (CoA) to comply with local regulatory requirements
- Consolidate all tests, instruments, sample information, and results data in one place
- Keep a defensible audit trail of all sample and test results.
Since the focus of ISO 17025 is on the operation and management of the laboratory, a well-configured LIMS can also make a significant contribution to the processes that underpin ISO 17025. These include:
- Laboratory organization and management structure
- Document control
- Standardization of test methods to ensure consistency
- Analytical methodology and procedures, including tests and calibration
- Maintenance of a clear, audit-trailed, searchable record of all samples and test results and reports issued
- Adherence to approved testing protocols for different products/programs
- Attainment of QA information necessary to investigate deviations and out-of-specification (OOS) results
- Maintenance of staff competency records for the assigned tasks
- Provision of a clear record and assignment system for corrective and preventative actions.
The LIMS can therefore provide a structure that enables management to make informed decisions regarding results and documentation of the data behind each decision for regulators.
Choosing a LIMS for cannabis testing
TEQ Analytical Laboratories achieved ISO/IEC 17025:2005 accreditation through the American Association for Laboratories Accreditation (A2LA). The laboratory (Figure 2) offers wide-ranging testing programs, utilizing techniques such as HPLC for potency testing and rapid polymerase chain reaction (PCR) for STEC and Salmonella analysis, and will soon add ICP/MS for heavy metals analysis. It uses the Matrix Gemini LIMS from Autoscribe Informatics (Reading, Berkshire, U.K.). The LIMS features graphical configuration capability, which provides a set of configuration tools to allow the creation and modification of any screen or workflow to meet the precise requirements of a laboratory.
 Figure 2 – Testing at TEQ Analytical Laboratories.
Figure 2 – Testing at TEQ Analytical Laboratories.With multiple workflows needed for the different testing regimes, the staff at TEQ staff was able to contribute significantly to the system configuration (Figures 3 and 4). The LIMS databases for cannabis testing and food testing are completely independent of each other. Of particular importance were the sample tracking and management, chain-of-custody, audit trail, and communications capabilities, since TEQ handles more than 30,000 samples/year with a two-day turnround time requirement for the majority of analyses.
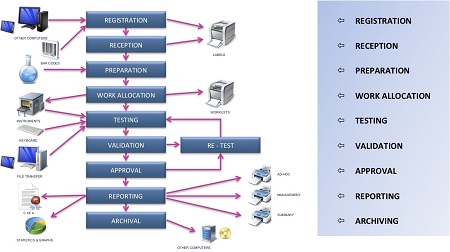 Figure 3 – Typical laboratory workflow.
Figure 3 – Typical laboratory workflow. Figure 4 – Typical LIMS laboratory process flow.
Figure 4 – Typical LIMS laboratory process flow.It is vital to know and understand exactly where samples are in the laboratory process and to ensure that results are not released before they have gone through all the necessary steps, such as QC approval, to safeguard against the possibility of results being released before they are verified. TEQ’s comprehensive quality assurance program includes an out-of-specification procedure in the event that results do not meet specifications provided by the client. Acceptable threshold limits for all tests are held within the LIMS, and if any test results lie outside of these limits they are automatically flagged by the system for review and follow-up by a member of the laboratory staff. The procedure also covers any occurrence that might affect test results; these events must be documented, tracked, and investigated to measure the effects on sample results and on the instrument, and then must be corrected to ensure they do not happen again. Certain OOS events can occur that will have little or no impact on sample results, but the lab and client must know this to ensure reliable analysis.
METRC compatibility
Metrc, a software application developed by Franwell (Lakeland, FL), is used by numerous states, including Colorado, to track, monitor, analyze, and guarantee compliance with complex regulations governing the growth, distribution, processing, and sale of cannabis products. Licensed testing facilities are tasked with timely and accurate entry of test results into Metrc. Using a third-party application programming interface, it is possible to automatically upload test results directly from the Matrix Gemini LIMS into Metrc. This saves time and eliminates the possibility of any data transcription errors.
Adaptability of LIMS
Configuration flexibility means that the LIMS can be modified as the requirements of the laboratory evolve to include additional testing or if regulatory requirements change. For example, TEQ will offer advanced testing for the presence of heavy metals once the Marijuana Enforcement Division begins certifying labs for this analysis. The LIMS can then be adapted to include these additional testing protocols.
Reference
- www.colorado.gov/pacific/enforcement/marijuanaenforcement
Seth Wong is president, TEQ Analytical Laboratories, 12635 E. Montview Blvd., Ste. 175, Aurora, CO 80045, U.S.A.; tel.: 303-577-6780; e-mail: [email protected]; www.teqanalyticallabs.com; Simon Wood is product manager, Autoscribe Informatics, U.K.; www.autoscribeinformatics.com