Featured Article

Please check out our Dynamic Mechanical Analyzer section for more information or to find manufacturers that sell these products.
Historically, dynamic mechanical analysis (DMA) measurements have focused on the analysis of materials as a function of an imposed temperature profile. However, most consumer products are subject to a variety of both temperature and relative humidity (RH) conditions during processing, storage, and end use. It is thus desirable to probe the combination of temperature and RH on the mechanical properties of materials to fully understand the environmental effects on structure and stability. DMA instruments often employ an inert purge to aid in temperature control and minimize thermal gradients. This purge effectively reduces the chamber relative humidity to very low levels, complicating the investigation of the effects of moisture or humid atmospheres on the mechanical behavior of materials.

Figure 1 - Q800 DMA and DMA-RH accessory.

Figure 2 - Operating range for the DMA-RH accessory.
The DMA-RH accessory, for use with the Q800 dynamic mechanical analyzer (Figure 1) (TA Instruments, New Castle, DE) is an integrated unit that includes the following major hardware components:
- The sample chamber mounts on the DMA in place of the standard furnace and completely encloses the sample. Peltier elements control the temperature to within ±0.1 °C. The DMA-RH sample chamber accommodates standard DMA clamps such as tension, cantilever, and three-point bending. The sample chamber is quickly and easily removed, allowing for rapid conversion back to the standard DMA furnace configuration.
- A heated vapor transfer line is maintained at a temperature above the dewpoint temperature of the purge gas in order to avoid condensation.
- The DMA-RH accessory cabinet contains the humidifier and associated electronics, which precisely controls temperature and humidity of the sample environment. The Q800 DMA communicates directly with the accessory and automatically controls all subsystems that are associated with generating and maintaining both temperature and humidity using Thermal Advantage instrument software.
The DMA-RH accessory provides a wide range of combined temperature and relative humidity, as shown in Figure 2. Table 1 provides the operating specifications. Operating modes include: isothermal/isohume, isothermal/step RH, isothermal/ramp RH, isohume/step temperature, isohume/ramp temperature, and step temperature/step RH. All of these operating modes are fully programmable using the Thermal Advantage software and associated method editor utility.
Results and discussion
Analysis of Nylon 6
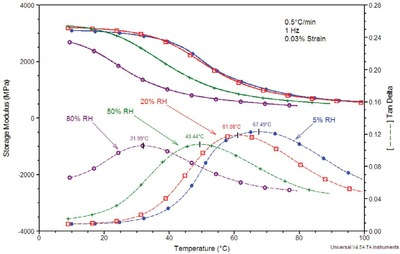
Figure 3 - Isohume DMA-RH analysis of Nylon 6.
Nylon 6 is strongly plasticized by water; as such the mechanical properties of this polymer will be dependent on the surrounding relative humidity.1 The data in Figure 3 illustrate the effect of relative humidity on the glass transitions of Nylon 6 as measured on the Q800 DMA equipped with the DMA-RH accessory. In this experiment, a sample was analyzed in single cantilever mode at a frequency of 1 Hz over the Tg temperature range at a variety of constant RH conditions. As expected, the mechanical properties are influenced significantly by the imposed relative humidity. The glass transition peak in tan δ is suppressed nearly 40 °C over an imposed relative humidity range of 75% RH.
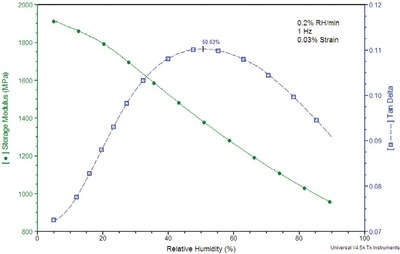
Figure 4 - Isothermal (35 °C) DMA-RH analysis of Nylon 6.
The versatility of the DMA-RH accessory also allows for the Nylon 6 sample to be analyzed under isothermal conditions, with an applied RH ramp. The data in Figure 4 contain the results from this experiment in which the sample was held isothermal at 35 °C while the humidity was ramped at 0.2% RH/min from 5 to 95% RH. Under these conditions, the Tg is identified by the peak in tan δ at 50.6% RH. The lower Tg temperature relative to the previous plot likely indicates diffusion effects or hysteresis in the sample.
Analysis of gelatin capsule material
Gelatin is a naturally occurring material that is used in a wide array of applications, most typically in foods, and in pharmaceutical, photographic, and technical products. The use of gelatin in the manufacture of various pharmaceutical dosage forms dates back to the early nineteenth century and possibly earlier.2 When stored in an ambient, low-humidity environment, gelatin is remarkably stable. However, when combined with water, gelatin forms a semisolid colloid gel, and this can profoundly affect its mechanical properties. Particularly as it relates to storage and handling conditions, it is important to understand and quantify these effects in situ.

Figure 5 - Relative humidity ramp of gelatin material at 25 °C and 40 °C.
The data in Figure 5 illustrate the effect of increasing relative humidity on a gelatin sample cut from the side wall of a two-piece capsule at both 25 °C and 40 °C. In each case, as the relative humidity is increased, the material undergoes a multistep transition, resulting in a significant decrease in modulus near 80% RH. The transition is resolved in both the modulus and tan δ signals. Pharmaceutical materials are often stored in bathrooms, which are typically the hottest and most humid locations in a dwelling. The combinations of temperature and RH can easily achieve those shown in the example, which could result in storage and stability concerns. These data suggest that even at ambient temperature, the storage conditions for gelatin capsules should be maintained below 70% RH to ensure mechanical integrity of the material.
Analysis of pasta product
Moisture content in cooked pasta is the primary factor affecting texture and mouth-feel, two properties that are directly related to the mechanical behavior of the material. The control of these parameters is of great interest to pasta manufacturers, since precooked products are becoming increasingly popular. It is known that a distinctive moisture distribution in pasta can be correlated to the so-called “al dente” state.3 The goal is to develop precooked pasta with an al dente texture similar to that of freshly prepared pasta. However, the texture of pasta deteriorates very rapidly after cooking and during distribution to stores. It is thus important to understand the combined effect of temperature and humidity on the pasta structure, to optimize the production, transportation, and storage stability, as well as to control the texture and end-use properties.

Figure 6 - RH cycle (60 °C) of fettuccine pasta.
The data in Figure 6 show the result of cycling RH on fettuccine pasta measured in a single cantilever at 60 °C. The RH was scanned from 0 to 95% and back to 0% at 0.08% RH/min (step-iso). It is clear from these results that the structure of the pasta material undergoes a significant softening near 70% RH. On increasing the RH, the relatively sharp tan δ peak suggests a rapid decrease in mechanical strength, which can be correlated to the onset of the al dente texture. Upon decreasing the RH, the transition exhibits reversibility; however, the material does not regain the original modulus of the dry pasta. This could be attributed to water retention in the structure, which plasticizes the material, or to an irreversible change in morphology, such as gelation or gelatinization of the starch molecules. The latter is more likely, as corresponding gravimetric sorption analysis data indicate complete reversibility in water uptake.
Analysis of Nafion® 112
Recent research4 has focused on alternative fuel technologies including Proton Exchange Membrane Fuel Cells (PEMFC), which contain polymeric membranes such as Nafion 112 (Nafion is a registered trademark of E.I. du Pont de Nemours and Co. [Wilmington, DE] or its affiliates). PEM properties can significantly change as functions of time and exposure to elevated temperatures and humidity, since water is the primary by-product of the electrochemical reaction of the fuel cell. Nafion is widely used in PEMFC technology developed for automotive applications, where the required operational range is from 80 °C to 110 °C. It has a high power density and is well known in the industry. It is understood that the Nafion membrane will experience various hydration levels within the fuel cell environment during operation. As such, stress relaxation experiments at different combinations of temperature and relative humidity can help characterize polymer swelling, and the resultant impact on mechanical characteristics.
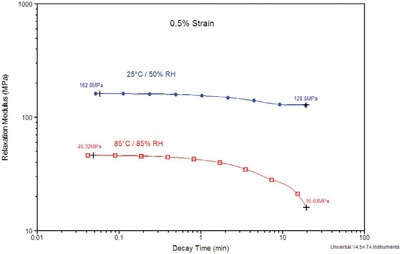
Figure 7 - Stress relaxation of Nafion 112 under varying conditions of temperature and RH.
The results in Figure 7 show the stress relaxation modulus of the Nafion 112 membrane at two different combinations of temperature and relative humidity. The upper blue curve was measured at 25 °C/50% RH, to simulate typical ambient conditions. The lower red curve was measured at 85 °C/85% RH, a more aggressive atmosphere nominally chosen as it represents an electronic materials standard. From the data in Figure 7, it is clear that the mechanical properties of the Nafion material are strongly influenced by the surrounding environment. Under the simulated ambient conditions, the relaxation modulus is relatively stable. Increasing the temperature and pressure effectively decreases the initial relaxation modulus by 4×. In addition, the time-dependence of the relaxation modulus is more profound, exhibiting a considerable decrease between 1 and 20 min, ultimately resulting in a relaxation modulus approximately 10× lower than that measured under ambient conditions.
Summary
The Q800 dynamic mechanical analyzer and DMA-RH accessory provide a wide range of temperature and relative humidity. The versatility of the accessory allows for programming of static or dynamic conditions of atmospheric control, enabling the mechanical analysis of a diverse spectrum of materials.
Please check out our Dynamic Mechanical Analyzer section for more information or to find manufacturers that sell these products.
References
- Jin, X.; Ellis, T.S.; Karasz, F.E. J. Poly. Sci.: Poly. Phy. Ed.1984, 22(10), 1701–17.
- www.gelatin-gmia.com/html/rawmaterials_app.html (Gelatin Manufacturer’s Institute of America).
- Horigane, A.K.; Naito, S.; Kurimoto, M.; Irie, K.; Yamada, M.; Motoi, H.; Yoshida, M. Cereal Chem.2005, 83(3), 235–42.
- a) Verbrugge, M.W.; Hill, R.F. J. Electrochem. Soc.1990, 137, 3370; b) Springer, T.E.; Zawodzinski, T.A.; Gottesfeld, S. J. Electrochem. Soc. 1991, 138, 2334; c) Wnek, G.E.; Ehrenberg, S.G.; Serpico, J.M.; Sheikh-Ali, B.M.; Tangredi, T.N.; Zador, E. Second International Symposium on New Materials for Fuel Cell and Modern Battery Systems, Canada, 1997; d) Uan-Zo- li, J.T. M.S. Thesis, Virginia Polytechnic Institute and State University, 2001.
Dr. Aubuchon is Thermal Analysis Product Manager, TA Instruments, 159 Lukens Dr., New Castle, DE 19720, U.S.A.; tel.: 302-427-4000; fax: 302-427-4001; e-mail: [email protected].